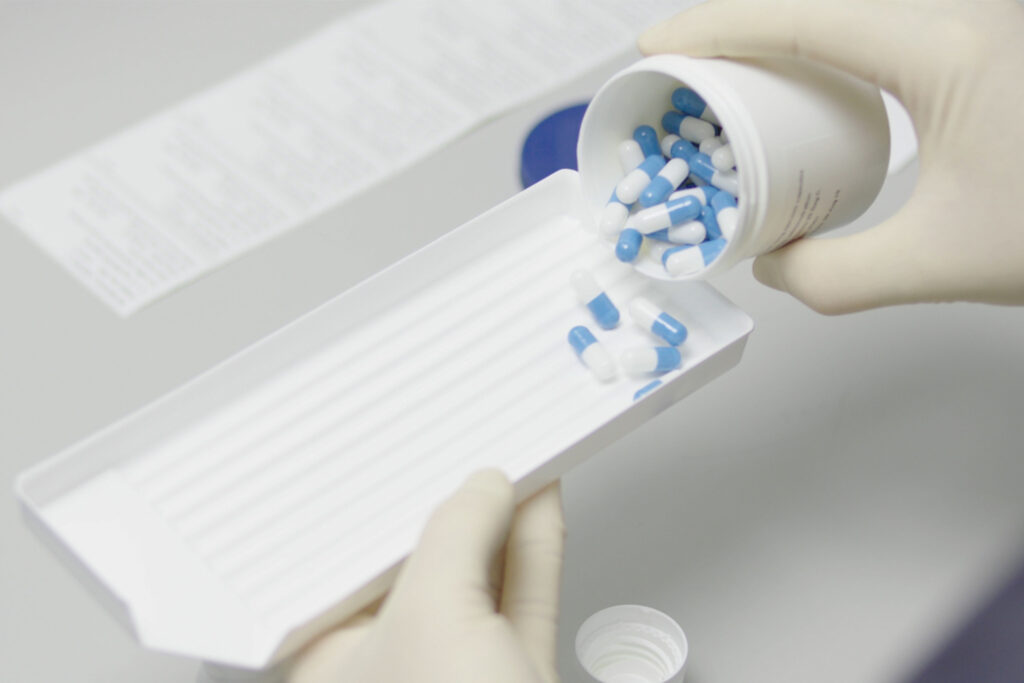
Simbec-Orion has years of experience conducting clinical trials of Investigational Medicinal Products (CTIMPs), or manufacturing, handling and managing GDP-compliant Investigational Medical Product (IMP) distribution to external sites. Our fully equipped IMP suite offers the capability to manufacture, package, QP certify, store and distribute your IMP.
If your clinical trial is being managed within our own Clinical Pharmacology Unit (CPU), you also benefit from the IMP suite being in the same building (and GCP accredited) – ensuring a compliant, efficient handover.
Find out more about our full clinical trial management services now.
EU IMP QP Certification & Release
Part of our ongoing strategy to provide fully integrated support to our clients includes the capability to import IMP into the EU, QP certify, and then release for use within clinical trial sites across the EU/EEA. This service forms part of our wider clinical trial management support for studies conducted by Simbec-Orion, but can also be delivered as a bespoke standalone service.
Most importantly, this capability includes direct-to-site import of radiopharmaceutical IMP to EU hospital sites for oncology trials, as well as the importation of a range of IMP formulations
Here, Christopher Jeans, QP, discusses the benefits this brings to our clients.


IMP Services
Our IMP management suite is able to handle compounding of various dosage forms:
- Sterile products for parenteral administration including intravenous (iv), intramuscular (im) and subcutaneous (sc)
- Non-sterile products such as: capsules, solutions, suspensions and topicals
- Radiopharmaceuticals
- Solutions for inhalation/nebulisation
Additionally, our IMP services can include:
- Primary and secondary packaging
- Double-blind randomisation and code break envelope production
- Annex 13 compliant IMP label design
- Storage (ambient, refrigerated, -20°C, -70°C)
- GDP-compliant IMP distribution to external sites
- Reconciliation, return and destruction of IMP
- Procurement of comparator products and NIMPs
Regulatory expertise and compliance
Our IMP management suite is GCP-compliant for efficient handovers of products from the IMP suite pharmacy to our in-house Clinical Pharmacology Unit (CPU).
We also adhere to full Good Distribution Practice (GDP) for external distribution of your IMP. This ensures the product is fully safe, compliant and ready for your study.
We can aid with compliance submissions and facilitate rapid contact with the appropriate Regulatory Health Authorities, Ethics Committees and Notified Bodies during the development and maintenance phase of your clinical trials.
Our Qualified Persons (QPs) can aid with declarations, the certification of IMP batches, and the compliant importation of IMPs into the UK.
Qualified Person services
IMP management services also include Qualified Person (QP) services. Our QPs have extensive experience in handling IMPs and can support your company’s goals – whether you’re a manufacturer or sponsor.
Under IMP management, our QP services include:
- QP certification of IMP batches
- QP Declarations
- Importation of IMP from outside of the UK
- Importation of IMP from an approved country (QP Oversight).
- IMPD writing and review
- Quality oversight
- CMC consultancy
- Supply chain audits
Handling IMPs
All IMP is handled within our access and temperature-controlled GMP facility, under the guidance of our Head of IMP Management.
The facility includes a Grade C environment for the manufacture of non-sterile IMP and a Grade A positive pressure isolator for the aseptic preparation of sterile IMP.
IMP Logistics
Simbec-Orion can help with the logistical planning of IMP management: the shipping and transport process, labelling and safe storage of the IMP. We ensure traceability and shipment visibility throughout the logistics process and ensure all logistics adhere to Good Distribution Practice (GDP).