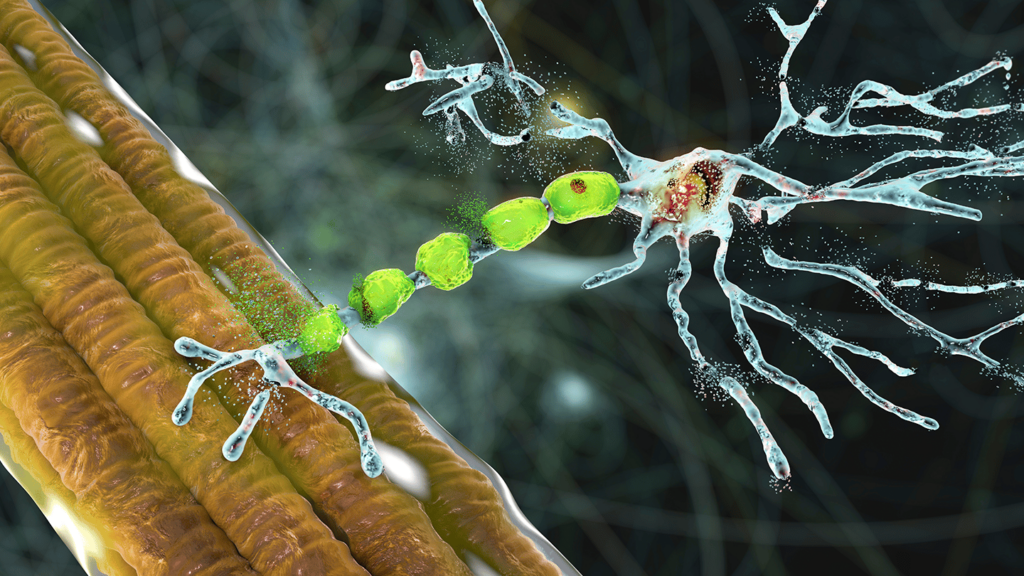
Amyotrophic lateral sclerosis (ALS) is a rare, progressive neurodegenerative disease that affects the motor neurons controlling voluntary muscle movement. The onset is usually quite gradual, with patients noticing muscle weakness – including tripping or dropping things, in the early stages. Not all patients with ALS experience the same symptoms, and progression rate and survival time can also vary. Diagnosis tends to be reached after a process of elimination, as several other treatable diseases have symptoms which mimic ALS, and there is no one test or procedure to give a definitive diagnosis. In the later stages of disease progression, ALS patients can expect to experience complete loss of mobility, including the ability to write, speak, walk and breathe.
Through medical research, the options available to patients living with ALS are improving. Simbec-Orion is currently supporting AL-S Pharma with their AP-101 Study in ALS, which is recruiting patients now.
AP-101 is a monoclonal antibody that binds to misfolded forms of SOD1, which are considered the most likely mechanism in ALS caused by SOD1 mutations and are also present in the CSF of sporadic ALS. The assumed mechanism of AP-101 is its specific binding to such misfolded SOD1 and its subsequent removal by phagocytosis, while not binding to the native form of SOD1 that is essential for human biological functions. AP-101 is administered by intravenous infusion.
In transgenic preclinical models, AP-101 generated highly significant therapeutic effects and the SAD study showed a very beneficial safety profile of AP-101 in humans at all dose levels tested.
Without any disease-modifying treatment modality, we believe that the current AP-101 phase 2 study offers a valuable treatment option for ALS patients by its multiple dosing during 6 months with an additional 6 months open-label extension.
Further information can be found on ClinicalTrials.gov here. If you are interested in taking part in the study, please contact us by email at [email protected]